During conversion/transformation, some energy gets wasted. But a team from MIT has created an electrochemical cell that can convert waste heat into electricity.
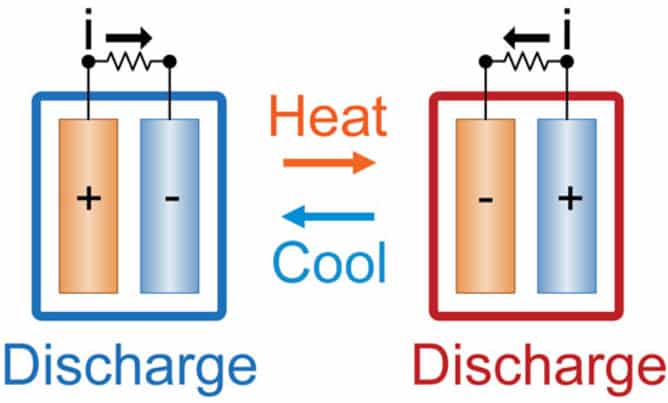
The electrochemical cell is made of Prussian blue nanoparticles and ferrocyanide and it uses different temperatures to convert heat to electricity. The electrochemical cell only needs low-grade waste heat – less than 100C – to charge batteries, and is a significant step forward compared to similar devices which either require an external circuit for charging or high temperature heat sources (300C).
At higher temperatures (60C), the cell was charged, and following cooling to 15C, the cell discharged energy. At lower temperatures the cell discharged more energy than was used to charge it, so converted heat to electricity. Note that, the most heat energy conversion is based on high temperature, and low-grade heat conversion devices will never be able to achieve high conversion efficiencies.
The amount of heat energy generated is dependent on the temperature and the Carnot limit. The Carnot limit is the maximum absolute amount of heat energy that can be converted to useful electricity. However, this first prototype can only convert 2% waste heat energy into electricity.
Anthony Vassallo, Delta Electricity Chair in Sustainable Energy Development at The University of Sydney, said, “It’s a great idea to be able to recover useful electrical energy from waste heat. While this will no doubt be improved, there are thermodynamic limits which basically say the maximum efficiency will always be low at the sort of temperatures these electrochemical cells could work at.”
The MIT researchers have publish their study in Proceedings of the National Academy of Science. Now they want to try use the technology to harvest heat from the environment in remote areas. But as solar arrays already dominate the market, and operate more efficiently, it is unlikely heat conversion technology will supercede them any time soon.
Source: The Conversation
[ttjad keyword=”t-mobile-contract-phone”]




document control software reviews [url=https://otvetnow.ru]https://otvetnow.ru[/url] how do you find the network security key
new york foreclosure attorneys [url=https://otvetnow.ru]https://otvetnow.ru[/url] credit card starting with 5
multiple sclerosis prevention [url=https://otvetnow.ru]https://otvetnow.ru[/url] hosted virtual desktop windows 7
web development courses online [url=https://otvetnow.ru]https://otvetnow.ru[/url] moving company cost estimate
what port does snmp use [url=https://otvetnow.ru]https://otvetnow.ru[/url] pest control in boston