Generators are normally big and heavy. The same goes for the hydrogen generator. But now, researchers at the US Department of Energy’s (DOE) Argonne National Laboratory have created a nano-sized hydrogen generator.
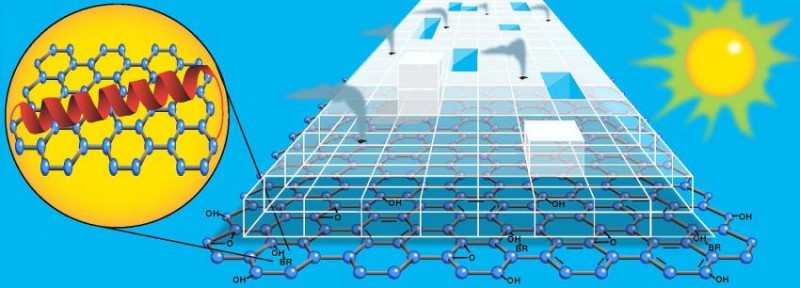
The nano-sized hydrogen generator uses light and a two-dimensional graphene platform to boost production of the hard-to-make element. In order to produce greater amounts of hydrogen using visible light, the researchers looked for a new material. The new material would need enough surface area to move electrons across quickly and evenly and boost the overall electron transfer efficiency. The researchers also needed a platform on which biological components, like bacteriorhodopsin (bR), could survive and connect with the titanium dioxide catalyst: in short, a material like graphene.
Graphene is a super strong, super light, near totally transparent sheet of carbon atoms and one of the best conductors of electricity ever discovered. Graphene owes its amazing properties to being two-dimensional. Elena Rozhkova, chemist at Argonne’s Center for Nanoscale Materials, a DOE Office of Science (Office of Basic Energy Sciences) User Facility said, “Graphene not only has all these amazing properties, but it is also ultra-thin and biologically inert. Its very presence allowed the other components to self-assemble around it, which totally changes how the electrons move throughout our system.”
Rozhkova’s mini-hydrogen generator works like this: both the bR protein and the graphene platform absorb visible light. Electrons from this reaction are transmitted to the titanium dioxide on which these two materials are anchored, making the titanium dioxide sensitive to visible light. Simultaneously, light from the green end of the solar spectrum triggers the bR protein to begin pumping protons along its membrane. These protons make their way to the platinum nanoparticles which sit on top of the titanium dioxide. Hydrogen is produced by the interaction of the protons and electrons as they converge on the platinum.
Examinations using a technique called Electron Paramagnetic Resonance (EPR) and time-resolved spectroscopy at the Center for Nanoscale Materials verified the movements of the electrons within the system, while electrochemical studies confirmed the protons were transferred. Tests also revealed a new quirk of graphene behavior.
Argonne postdoctoral researcher Peng Wang said, “The majority of the research out there states that graphene principally conducts and accepts electrons. Our exploration using EPR allowed us to prove, experimentally, that graphene also injects electrons into other materials.”
However, this nano-sized hydrogen generator proves that nanotechnology, merged with biology, can create new sources of clean energy. Researchers’ discovery may provide future consumers a biologically-inspired alternative to gasoline.
Source: Argonne National Laboratory
[ttjad keyword=”sony-ericsson”]