Sonitus Medical receives European CE mark for SoundBite hearing system and European mark indicates compliance with globally recognized quality management and assurance systems.SoundBite is a new hearing aid that transmits sound through a person’s teeth and directs sound through the jawbone and into the inner ear…….
Sonitus Medical Inc., a medical device company that manufactures the world’s first non-surgical and removable hearing prosthetic to transmit sound via the teeth announced that it has received CE Mark certification for its SoundBite Hearing System. The company has received its EC Certificate, which indicates Full Quality Assurance System Approval. This is the first European regulatory certification for the company and the product.“Receiving the CE Mark for our SoundBite Hearing System is a significant milestone toward the global commercialization of our non-surgical bone-conduction hearing device,” said Amir Abolfathi, CEO of Sonitus Medical. “We intend to pursue the process of reimbursement registration in the key EU countries in the coming months and then gain initial European clinical experience with the product in partnership with a limited number of Otologists, ENTs, and Acousticians in the EU.”Apart from the FDA clearance attained by the company earlier this year, CE Marking indicates that Sonitus Medical’s design and manufacturing processes for the SoundBite Hearing System comply with certain manufacturing and design standards under the European directive concerning medical devices. This certification enables Sonitus Medical to commercialize the SoundBite Hearing System in Europe.
ScreenShots :
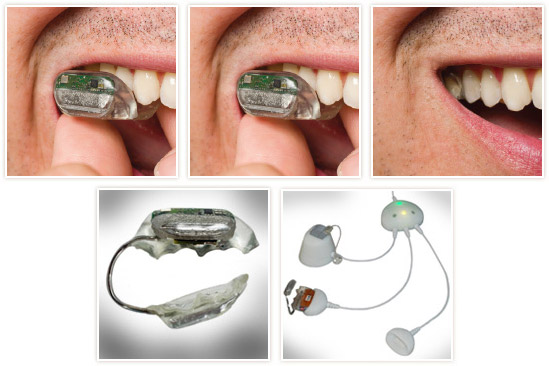

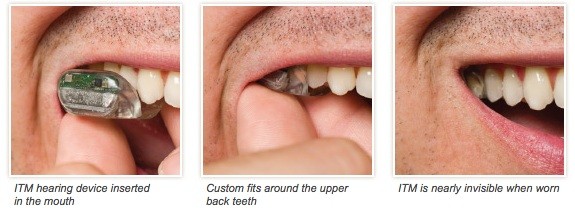

This is a very interesting invention.. Hats off to the inventors who
think about it in such a great manner.. I was wondering to know after
fixing it in the mouth.. Can anyone eat something or not?
durable medical equipment